From the beginning of 1918 until the end of 1920, the world suffered through a deadly influenza (flu) pandemic that infected an estimated 500 million people in numerous countries throughout the world. Before it was over, at least 50 million people had died, including 650,000 Americans. Nearly half of the U.S. soldiers who perished in Europe during World War I died from the influenza virus rather than from combat injuries. The rapid spread of the so-called “Spanish flu” has been attributed to closely packed troop quarters and massive troop movements, coupled with the increased travel possibilities available from the developing transportation system.
Although later influenza outbreaks have not matched the severity or devastation of the 1918-1920 pandemic, the number of influenza-related deaths in the United States in any given year is between 3,000 and 49,000, according to the U.S. Centers for Disease Control and Prevention (CDC). This wide range of death rates reflects circulation variability of numerous influenza virus strains in addition to wide fluctuations in duration and severity of each flu season. Regardless of the exact number of deaths in any recent year, the risk posed by new influenza outbreaks remains a serious threat to public health – and, for that reason, several government agencies (such as the CDC) are not only focusing greater attention on the strategies needed to mitigate the impact of any and all future flu pandemics, but also striving to ensure that the global death toll suffered in the early 20th century is not repeated.
Legislation & Guidelines for Outbreak Response
Recognizing that new epidemics and/or pandemics may arise before treatments that address specific diseases are cleared through the U.S. Food and Drug Administration (FDA), several steps already have been taken to ensure that the nation can expeditiously and effectively respond to such outbreaks. Following are three prominent examples of the actions already taken:
- President William J. Clinton’s 30 September 1999 Executive Order (EO) 13139 stipulates the conditions under which a promising, yet unlicensed, medical countermeasure may be administered to military personnel when no FDA-approved medical product is available.
- The Project BioShield Act of 2004 (Public Law 108-276; “An Act”) established the comprehensive Emergency Use Authorization (EUA) program, which permits the FDA to approve the emergency use of drugs, devices, and medical products that had not been previously approved, cleared, or licensed by the FDA.
- The U.S. Department of Defense (DOD) 27 February 2008 Instruction 6200.02 provides guidance that should be used on the application of FDA rules to DOD Force Health Protection Programs – specifically including directions on when it is appropriate to use an unapproved product under an EUA or, if an EUA is not feasible, under an investigational new drug (IND) application.
Enhancing the Nation’s Response
Beyond the toll on the U.S. civilian population, it has been estimated that another flu outbreak similar to the 2009 H1N1 pandemic could cause overall case (“attack”) rates exceeding 10 percent per month on U.S. military forces. The impact on military medical and operational capabilities could be considerable. For that reason, President Obama directed the DOD leadership to expand its Chemical Biological Defense Program portfolio by including the medical countermeasures needed to cope with emerging infectious diseases such as naturally occurring and/or biologically engineered influenza viruses. This guidance was provided in the 2009 National Security Council: President’s Policy Directive-2 (PPD-2): National Strategy for Countering Biological Threats.
DOD already takes a proactive role in providing seasonal flu vaccines to military personnel (and to all eligible beneficiaries). However, although the use of vaccines is considered one of the best ways to prevent infection, the effectiveness of a specific flu vaccine depends primarily on matching that vaccine to the strains that are forecasted for a given season. Because of the rapid mutation rate of the influenza virus prevalent in any given year, it is always a major challenge ensuring the prediction for the “next” virus. Moreover, those suffering from immune deficiencies, or allergies to egg products, cannot receive these vaccines. Reasons such as these have made it clear over the past 10 years that medical interventions for influenza should include not only vaccines but also post-exposure therapeutics.
Leading the effort to complement the DOD’s vaccine approach is the Joint Project Manager-Transformational Medical Technologies (JPM-TMT), Emerging Infectious Diseases-Influenza Medical Countermeasure (EID-Flu MCM) acquisition program. JPM-TMT is a component of the DOD’s Joint Program Executive Office for Chemical and Biological Defense (JPEO-CBD). The specific goals of JPM-TMT’s EID-Flu MCM acquisition program are: (a) to protect the nation’s warfighters from emerging infectious diseases – specifically from the influenza virus (whether naturally occurring or biologically engineered); and (b) to revolutionize influenza treatment by developing a novel, broad-spectrum (i.e., efficacious against multiple strains of influenza), post-exposure therapeutic.
Although anti-viral drugs are already currently available, the influenza virus is constantly mutating, and continues to demonstrate increased drug resistance. In the United States, four antiviral drugs are currently FDA-approved for use against influenza: amantadine and rimantadine – approved for influenza A; and Tamiflu® (oseltamivir phosphate) and Relenza® (zanamivir) – approved for both influenza A and influenza B. The CDC still recommends Tamiflu and Relenza for the treatment of influenza infection, but no longer recommends amantadine and rimantadine because of resistance that has built up against these two drugs. However, for many strains of the influenza virus, there are no FDA-approved therapeutics.
A New Paradigm to Flu Treatment Emerges
Recognizing that a new approach to fighting influenza should be taken, in May 2011, JPM-TMT’s EID-Flu MCM acquisition program issued a Request for Proposal (RFP) for drug candidates that: (a) were broad-spectrum; (b) had already filed an Investigational New Drug application with the FDA; (c) were not on clinical hold; and (d) demonstrated a unique mechanism of action. In March 2012, the program contracted with MediVector Inc. to further develop Favipiravir (T-705), a promising broad-spectrum drug candidate capable of addressing multiple strains of the influenza virus. T-705 demonstrated effectiveness against drug-resistant strains of influenza virus, including those resistant to Tamiflu®, Relenza®, amantadine, and rimantadine.
MediVector Inc. was selected through a full and open competition and an extensive source selection process by a team of interagency participants. T-705, which was initially developed by Toyama Chemical Co., LTD., a subsidiary of FUJIFILM Holdings Corporation, has undergone clinical trials in Japan that show high efficacy against influenza. The EID-Flu MCM contract with MediVector Inc. will help support the steps necessary to acquire FDA approval, which is required for such drugs in the United States.
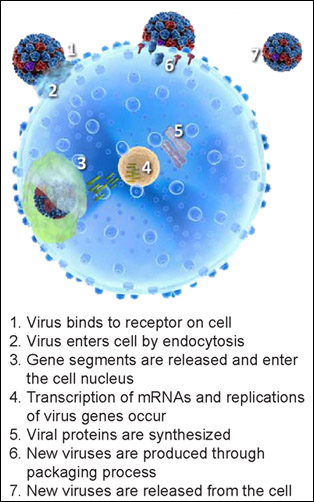
T-705’s unique mechanism of action (i.e., the specific biochemical interaction through which a drug substance produces its pharmacological effect) sets it apart from current FDA-approved anti-influenza drugs. The compound terminates the infection cycle of the flu by targeting and interfering with the viral proteins involved in the virus transcription and replication process. To illustrate the mechanism of action in T-705, it is helpful to understand how a virus infects a cell. Figure 1 demonstrates the influenza infection replication process in a cell and the subsequent spread or release of the virus from the cell.
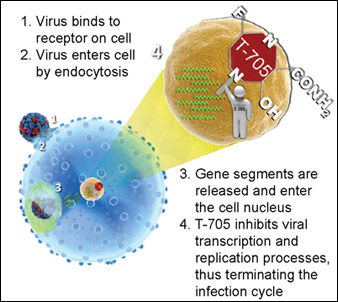
Figure 2 shows: (a) how the virus enters the cell in the usual fashion; and (b) how application of T-705 disrupts the infection replication process. Results from a 2005 study (Furuta et al.) – published in Antimicrobial Agents and Chemotherapy by the American Society for Microbiology – indicate that “T-705 interferes with an early to middle stage of influenza virus replication.”
Even more valuable is T-705’s demonstrated effectiveness against viruses with similar replication processes – including some on the CDC’s list of Category A and B bioterrorism agents. These results are particularly important in demonstrating how the DOD’s efforts in leveraging existing research and development projects help to fight other viruses that pose bioterrorism risks.
To briefly summarize: Through JPM-TMT’s strategic investments and partnerships with innovative biotech firms, pharmaceutical corporations, other government agencies, and academic institutions, DOD is taking an active role in expanding the nation’s medical inventory of the therapeutics needed to cope with emerging infectious diseases. Providing support for the nation’s warfighters is the program’s primary focus, of course; success – in the form of an FDA-approved, broad-spectrum counter-influenza drug – will benefit the nation as a whole. Moreover, by facilitating the advanced development and acquisition of broad-spectrum medical countermeasures and systems, JPM-TMT also enhances the DOD’s, and the nation’s, ability to respond to and cope with potential biowarfare agents and a future national infectious-disease emergency or pandemic.
____________
For additional information on: JPM-TMT, visit www.jpmtmt.mil
Yousuke Furuta et al.’s 2005 study “Mechanism of action of T-705 against influenza virus” (published in Antimicrobial Agents and Chemotherapy, Vol. 49, 981-986), visit https://journals.asm.org/doi/full/10.1128/aac.49.3.981-986.2005

David Gibson
Lieutenant Colonel David Gibson is the Product Manager for Joint Project Manager-Transformational Medical Technologies (JPM-TMT), Emerging Infectious Diseases-Influenza Medical Countermeasure acquisition program (EID-Flu MCM). JPM-TMT is a component of the Joint Program Executive Office for Chemical and Biological Defense (JPEO-CBD). The mission of JPM-TMT is to provide the warfighter and the nation with innovative medical solutions to protect against and treat emerging, genetically engineered, or unknown biological threats. Lieutenant Colonel Gibson has previously served in a variety of command and staff positions. He was last assigned to the Pentagon as the Executive Assistant to the Army Surgeon General and Commanding General, U.S. Army Medical Command.
- This author does not have any more posts.






